|
One candidate sensor is GCN2, a tRNA-binding protein How cells sense amino acid concentration is not fully understood. Leucine has a dominant effect on skeletal muscle and heart ( Mortimore and Pösö 1987). However, such profiles depend on cell type because amino acid metabolism differs greatly among tissues. Leu, Tyr, Phe, Gln, Pro, His, Trp, Met,Īnd Ala suppress autophagy in ex vivo perfused liver ( Mortimore and Pösö 1987). In many types of cultured cells, but the effects of individual amino acids differ. Depletion of total amino acids strongly induces autophagy In mammals, regulation of autophagy appears to be highly complicated. Nitrogen or carbon starvation also triggers autophagy in plant cells ( Moriyasu and Ohsumi 1996 Yoshimoto et al. In yeast, nitrogen starvation is the most potent stimulus, but withdrawal of other essential factors such as carbon,Īuxotrophic amino acids and nucleic acids, and even sulfate can induce autophagy, albeit less efficiently ( Takeshige et al. The most typical trigger of autophagy is nutrient starvation in this sense, lack of any type of essential nutrient can induceĪutophagy. Previous Section Next Section The process of autophagy Induction of autophagy Thus, in this review, I will attempt to follow the role of autophagy in a process-based manner. These step-dependent functions may allow autophagy to be multifunctional. To exert different functions in a variety of cellular contexts. However, even this distinction is too simplified and cannot be applied to more complicated issues.Īutophagy consists of several sequential steps: sequestration, degradation, and amino acid/peptide generation. The former is used to produce amino acids following starvation, while the latter is important for constitutive turnover To understand the various roles of autophagy, it may be useful to subclassify macroautophagy into “induced autophagy” and In vivo autophagic activity or total proteolysis is not sustained, and decreases during prolonged starvation ( Mortimore et al. However, the range of autophagy seems to be strictly controlled The toxicity of autophagy, particularly observed in cultured cells. This may explain many of the controversial issues related to Too muchĭestruction without construction would be harmful for cells. How can a bulk degradation system exert so many functions? One determinant is probably the extent of degradation. Therefore, it may be difficult to draw simplified connections between autophagy Life?” Of course, inflammation is required for the anti-bacterial response, but the inflammatory response associated withīacterial pneumonia can be life-threatening. This is like the question: “Is inflammation good or bad for To generalize the role of autophagy in cancer and cell death. Additionally, in some situations, the contribution of autophagy seems to be very complicated. 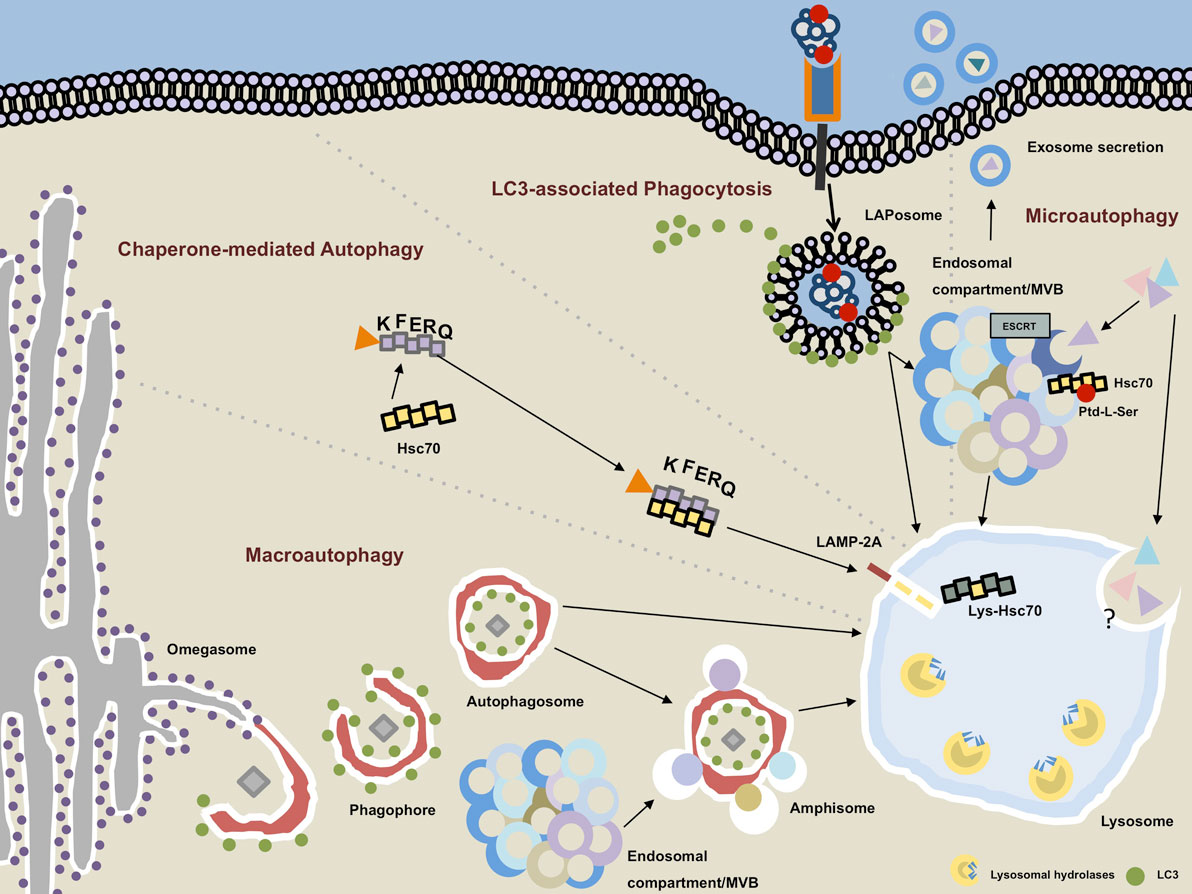
Roles than expected, such as starvation adaptation, intracellular protein and organelle clearance, development, anti-aging,Įlimination of microorganisms, cell death, tumor suppression, and antigen presentation ( Mizushima 2005). However, recent studies have clearly demonstrated that autophagy has a greater variety of physiological and pathophysiological To assume that the ubiquitin–proteasome system has numerous specific functions because it can selectively degrade thousands System, which specifically recognizes only ubiquitinated proteins for proteasomal degradation. This feature is in marked contrast to the ubiquitin–proteasome As autophagosomes engulf a portion of cytoplasm,Īutophagy is generally thought to be a nonselective degradation system. Autophagy is mediated by a unique organelle called the autophagosome. Usually indicates macroautophagy unless otherwise specified ( Fig. There are three types of autophagy-macroautophagy, microautophagy, and chaperone-mediated autophagy-and the term “autophagy” This process is quite distinct from endocytosis-mediated lysosomal degradation of extracellular and plasma membrane proteins. Is summarized, and the role of autophagy is discussed in a process-based manner.Īutophagy is a general term for the degradation of cytoplasmic components within lysosomes ( Cuervo 2004 Levine and Klionsky 2004 Shintani and Klionsky 2004 Klionsky 2005, 2007 Mizushima and Klionsky 2007). Autophagy consists of several sequential steps-sequestration, transport to lysosomes, degradation,Īnd utilization of degradation products-and each step may exert different function. Recent progress has demonstrated that autophagy plays a wide variety of physiological and pathophysiological roles, whichĪre sometimes complex. Autophagy is an intracellular degradation system that delivers cytoplasmic constituents to the lysosome.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
